|
3/20/2023 0 Comments Albuterol 90 mcg 
This field indicates whether the product has been removed/excluded from the NDC Directory for failure to respond to FDA"s requests for correction to deficient or non-compliant submissions ("Y"), or because the listing certification is expired ("E"), or because the listing data was inactivated by FDA ("I"). This is the date when the listing record will expire if not updated or certified by the product labeler.Įxclude Flag What is the NDC Exclude Flag? Listing Expiration Date What is the Listing Expiration Date? This is the date that the labeler indicates was the start of its marketing of the drug product. Start Marketing Date What is the Start Marketing Date? NDA - A product marketed under an approved New Drug Application. The complete list of codes and translations can be found at under Structured Product Labeling Resources. Currently, only final marketed product categories are included. One and only one Marketing Category may be chosen for a product, not all marketing categories are available to all product types. Product types are broken down into several potential Marketing Categories, such as NDA/ANDA/BLA, OTC Monograph, or Unapproved Drug. Marketing Category What is the Marketing Category? For unapproved drugs, this field will be null. If the designated Marketing Category is OTC Monograph Final or OTC Monograph Not Final, then the Application number will be the CFR citation corresponding to the appropriate Monograph (e.g. This corresponds to the NDA, ANDA, or BLA number reported by the labeler for products which have the corresponding Marketing Category designated. Name of Company corresponding to the labeler code segment of the Product NDC.įDA Application Number What is the FDA Application Number? Product Labeler Information What is the Labeler Name? 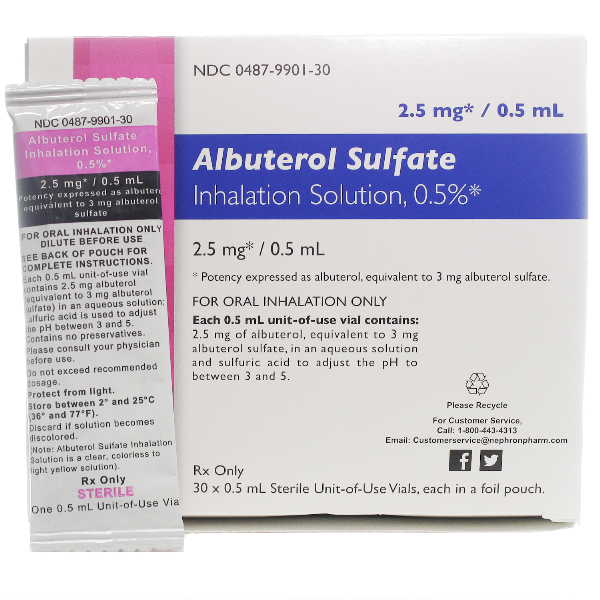
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
